FDA: Electroshock has risks but is useful to combat severe

FDA: Electroshock has risks but is useful to combat severe

After FDA bans Judge Rotenberg Center from using electric shock devices, advocates seek public apology, reparations

FDA bans electric shock devices for conditioning against aggressive behaviors

Interventional Treatment Options for Patients: ECT, VNS, and TMS

Watchdog Group CCHR Seeks to Stop American Psychiatric Association's Push to Reclassify Electroshock Devices for Use on Children

Can electrical brain stimulation boost attention, memory, and more? - Harvard Health

NRx Pharmaceuticals Reports Minutes of Recent U.S. Food and Drug Administration (FDA) Meeting on the Development of NRX-101 to Treat Severe Bipolar Depression in Patients with Suicidality

Judge Rotenberg Educational Center: Federal appeals court vacates FDA rule banning electric shock devices to treat self-harming behavior
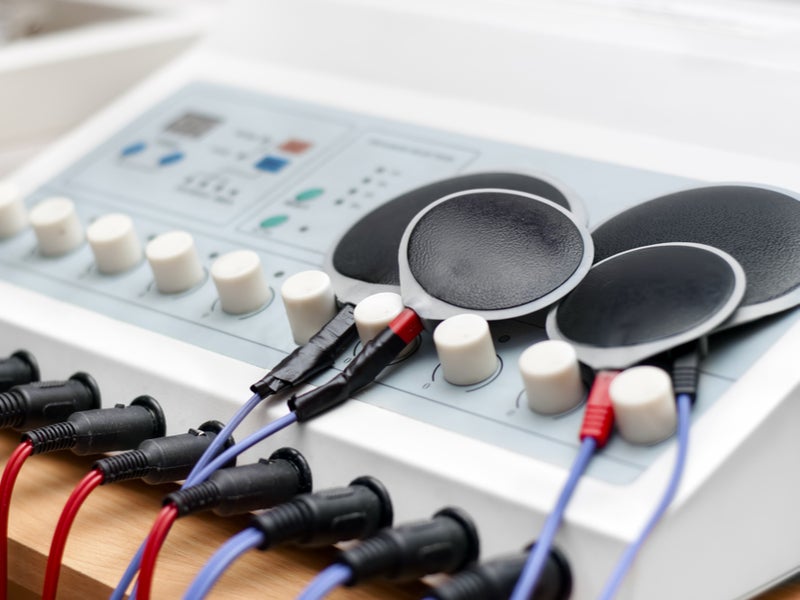
US ESDs ban: the rise and fall of electroshock aversion therapy

FDA considers ban on electric shock conditioning for autistic patients, US news
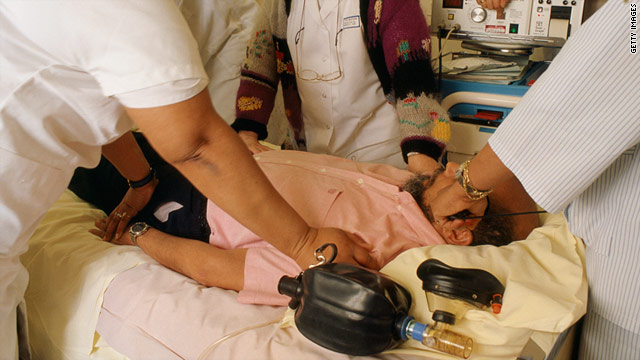
FDA panel deems electroshock devices high risk

7 of the Most Outrageous Medical Treatments in History

FDA unjustified in downgrading shock therapy brain injury risks - Washington Times


